烟草野火病叶际微生物群落结构多样性与碳源代谢表征
摘要:
为明确感染烟草野火病叶片结构及叶际微生物形态、碳源代谢能力和群落结构多样性特征,分别采用扫描电镜、BIOLOG ECO和Illumina NovaSeq高通量测序技术研究了感病烟叶与健康烟叶的叶片结构、叶际细菌和真菌群落结构的差异。结果表明,感病烟叶表面附着大量细菌、真菌及菌丝和孢子,烟草叶片受损严重。感病烟叶叶际微生物群落对羧酸类物质(丙酮酸甲酯和D-苹果酸等)的利用能力最强,健康烟叶叶际微生物群落对聚合物(Tween-40和Tween-80等)的利用能力最强,两组烟叶叶际微生物群落均不能高效利用酚类化合物(2-羟基苯甲酸和4-羟基苯甲酸)。感病和健康烟叶叶际细菌和真菌在门水平上差异不显著,但在属水平上差异显著。其中,感病烟叶的优势细菌属为假单胞菌属(Pseudomonas,67.43%)和泛菌属(Pantoea,5.75%),健康烟叶的优势细菌属为劳尔氏菌属(Ralstonia,17.62%)、假单胞菌属(15.79%)和反刍杆菌属(Ruminobacter,12.76%);感病烟叶的优势真菌属为茎点霉属(Phoma,24.68%)、链格孢属(Alternaria,22.87%)和Phialocephala(5.72%),健康烟叶的优势真菌属为枝孢属(Cladosporium,14.80%)、Sampaiozyma(8.19%)和被孢霉属(Mortierella,5.23%)。感病烟叶和健康烟叶叶际细菌和真菌的多样性、丰富度等均存在显著差异,暗示着烟草野火病的发生存在假单胞菌等细菌与茎点霉等真菌的复合侵染。
Abstract:
To clarify the characteristics of leaf structure and phyllosphere microbial morphology, carbon source metabolic capacity, and community structure diversity for tobacco leaves affected by wildfire disease, scanning electron microscopy, BIOLOG ECO, and Illumina NovaSeq high-throughput sequencing technologies were used to investigate the differences in leaf structure, phyllosphere bacteria and fungal communities between infected and healthy tobacco leaves. The results showed that a significant number of bacterial hyphae, fungal hyphae, and spores adhered to the surfaces of infected tobacco leaves that were severely damaged. The phyllosphere microbial communities of the infected tobacco leaves had the highest utilization capacity for carboxylic acids (methyl pyruvate and D-malate, etc.), whereas those of healthy tobacco leaves had the highest utilization capacity for polymers (Tween-40 and Tween-80, etc.). Both leaf groups were inefficient in the utilization of phenolic compounds (2-hydroxybenzoic acid and 4-hydroxybenzoic acid). Although no significant differences were observed in the phyllosphere bacterial and fungal communities between the infected and healthy tobacco leaves at phylum level, significant differences were observed at the genus level. Dominant bacterial genera of the infected tobacco leaves were Pseudomonas (67.43%) and Pantoea (5.75%), whereas Ralstonia (17.62%), Pseudomonas (15.79%) and Ruminobacter (12.76%) dominated in the healthy samples. Dominant fungal genera were Phoma (24.68%), Alternaria (22.87%) and Phialocephala (5.72%) on the infected tobacco leaves, whereas Cladosporium (14.80%), Sampaiozyma (8.19%) and Mortierella (5.23%) on the healthy tobacco leaves. Significant differences in diversity and richness between phyllosphere bacterial and fungal communities of the infected and healthy tobacco leaves indicated that a composite infection of bacteria such as Pseudomonas spp. and fungi such as Phoma spp. contributed to the progression of tobacco wildfire disease.
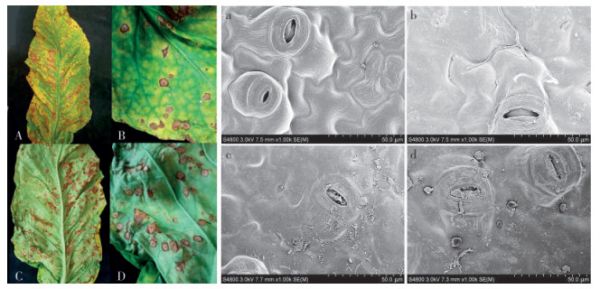
图 1 不同症状烟叶的形态扫描电镜观察与特征比较
A和B为感病烟叶正面危害症状,C和D为感病烟叶背面危害症状;a为健康烟叶表面电镜组织形态,b、c、d均为感病烟叶表面组织形态。
Fig. 1 Morphological observation by scanning electron microscopy and characterization of tobacco leaves with different symptoms
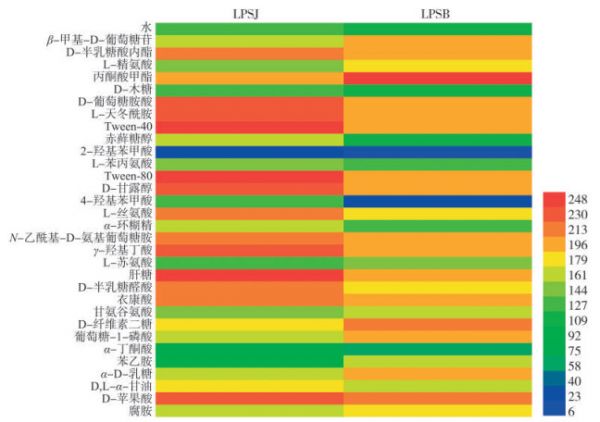
图 2 健康烟叶与感病烟叶叶际微生物的碳源代谢图谱
图例颜色表示Biolog OmniLog R系统AWCD值的大小。
Fig. 2 Carbon source metabolism map of phyllosphere microorganisms of infected and healthy tobacco leaves
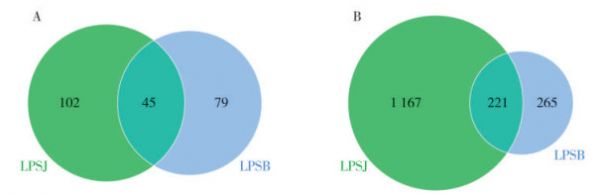
图 3 健康(LPSJ)和感病(LPSB)烟叶样品细菌(A)和真菌(B)ASV分布Venn图
Fig. 3 Venn diagrams of ASV distribution for bacteria (A) and fungi (B) in healthy (LPSJ) and infected (LPSB) tobacco leaf samples
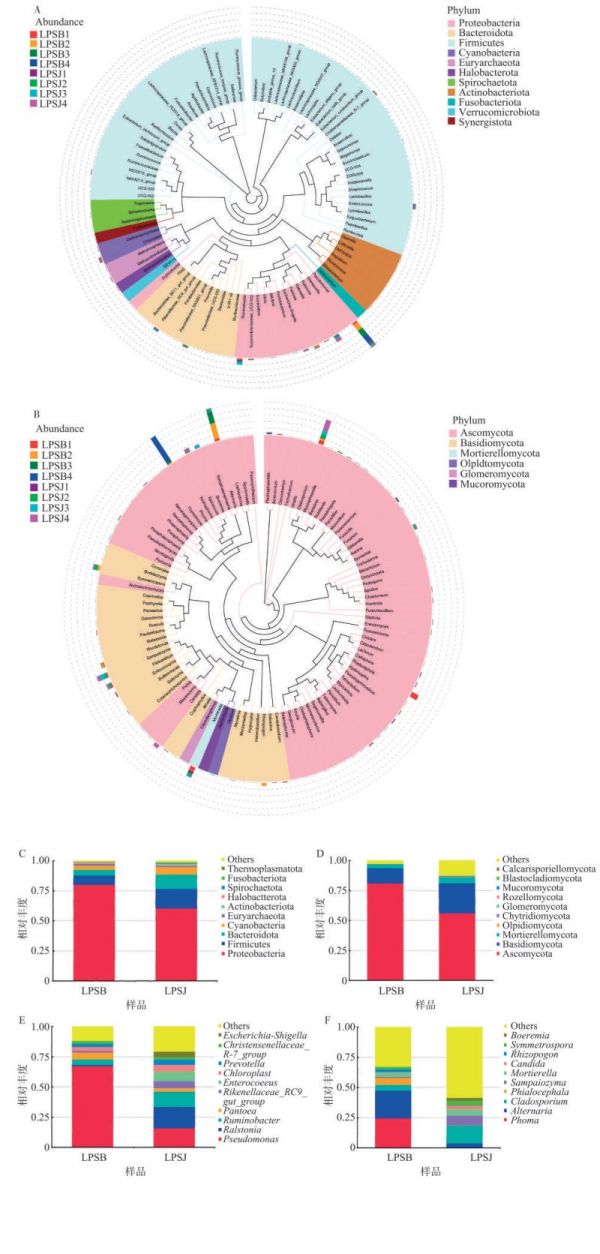
图 4 健康与感病烟叶叶际细菌(A)、真菌(B)物种进化树与细菌门(C)、属(E)和真菌门(D)、属(F)水平组间的相对丰度
A、B左侧图例为样本信息,右侧图例为属水平物种对应的门水平的分类信息;图C、D、E、F图例中Others表示图中10个门/属之外的其他所有门/属的相对丰度之和。
Fig. 4 Evolutionary trees of phyllosphere microorganisms, bacteria (A) and fungi (B), on healthy and infected tobacco leaves, and relative abundances of bacterial phylum (C), bacterial genus (E), fungal phylum (D) and fungal genus (F) groups
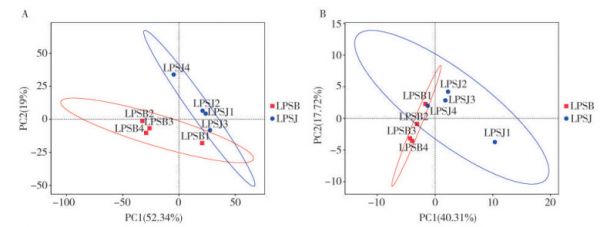
图 5 健康(LPSJ)和感病(LPSB)烟叶样本中细菌(A)与真菌(B)群落的主成分分析(PCA)
横坐标表示第一主成分,纵坐标表示第二主成分,百分比表示主成分对样本差异的贡献值。
Fig. 5 Principal component analysis (PCA) of bacterial (A) and fungal (B) communities of healthy (LPSJ) and infected (LPSB) tobacco leaf samples
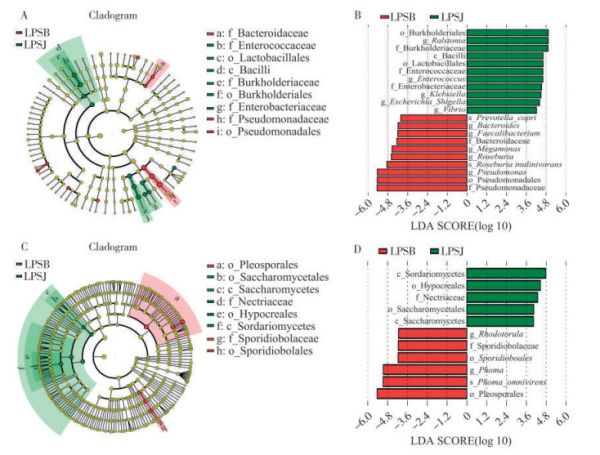
图 6 细菌与真菌群落组间进化分支图与LDA值分布柱形图
A.细菌进化分支图;B.细菌LDA分布图;C.真菌进化分支图;D.真菌LDA分布图。在进化分支图中,由内至外辐射的圆圈代表了由门至属(或种)的分类级别。在不同分类级别上的每一个小圆圈代表该水平下的一个分类,小圆圈直径大小与相对丰度大小呈正比。LDA值分布柱状图中展示了LDA score大于设定值(默认设置为4)的物种,即组间具有统计学差异的Biomarker。
Fig. 6 Intergroup evolutionary branching diagrams of bacterial and fungal communities and bar charts of LDA value distribution
表 1 不同烟叶样品的序列数据
Tab. 1 Sequence data of different tobacco leaf samples
样品 有效序列/条 碱基数/nt 平均长度/nt 细菌 真菌 细菌 真菌 细菌 真菌 LPSB1 76 812 55 540 31 194 278 14 315 321 406 258 LPSB2 82 984 65 834 33 823 340 15 949 946 408 242 LPSB3 60 323 72 532 25 132 199 15 306 587 417 211 LPSB4 75 970 64 386 31 145 999 13 647 568 410 212 LPSJ1 70 754 64 143 28 782 147 17 015 930 407 265 LPSJ2 83 541 70 913 33 943 411 17 923 390 406 253 LPSJ3 68 900 68 345 28 034 166 17 345 137 407 254 LPSJ4 85 154 69 099 34 582 234 15 719 312 406 227表 2 样品组间α多样性指数①
Tab. 2 Alpha diversity index between sample groups
样品 Shannon指数 Simpson指数 Chao1指数 Pielou_e指数 覆盖度 细菌 LPSJ 4.28±0.90a 0.89±0.06a 58.89±31.49a 0.77±0.43a 0.982 LPSB 2.95±1.19a 0.70±0.18a 71.69±39.84a 0.55±0.22a 0.963 真菌 LPSJ 6.10±1.45b 0.94±0.06a 480.99±180.07b 0.69±0.12b 1.000 LPSB 2.78±1.77a 0.61±0.30a 159.03±99.83a 0.38±0.20a 1.000注:①表中数据为平均值±标准差,同列不同字母表示有显著性差异(P<0.05)。 [1]Xin X F, Kvitko B, He S Y. Pseudomonas syringae: What it takes to be a pathogen[J]. Nature Reviews Microbiology, 2018, 16(5): 316-328.
[2] 陈瑞朋, 卢凯, 张敏, 等. 烟草野火病菌特异性检测引物筛选及应用[J]. 中国烟草科学, 2018, 39(1): 72-76.CHEN Ruipeng, LU Kai, ZHANG Min, et al. Screening and application of Pseudomonas syringae pv. Tabaci-specific primers[J]. Chinese Tobacco Science, 2018, 39(1): 72-76.
[3]Kusstatscher P, Cernava T, Harms K, et al. Disease incidence in sugar beet fields is correlated with microbial diversity and distinct biological markers[J]. Phytobiomes Journal, 2019, 3(1): 22-30. doi: 10.1094/PBIOMES-01-19-0008-R
[4] 刘畅, 汪汉成, 谢红炼, 等. 感染赤星病烟草叶际细菌的多样性分析[J]. 烟草科技, 2020, 53(2): 8-14. doi: 10.16135/j.issn1002-0861.2019.0114LIU Chang, WANG Hancheng, XIE Honglian, et al. Biodiversity analysis of phyllosphere bacterial genus from tobacco leaves infected by brown spot disease[J]. Tobacco Science & Technology, 2020, 53(2): 8-14. doi: 10.16135/j.issn1002-0861.2019.0114
[5] 黄宇, 汪汉成, 陈乾丽, 等. 感染白粉病烟株典型病级叶际真菌群落结构与多样性分析[J]. 烟草科技, 2021, 54(4): 8-14. doi: 10.16135/j.issn1002-0861.2019.0475HUANG Yu, WANG Hancheng, CHEN Qianli, et al. Community structure and diversity of phyllosphere fungi in tobacco plants with typically infected tobacco powdery mildew[J]. Tobacco Science & Technology, 2021, 54(4): 8-14. doi: 10.16135/j.issn1002-0861.2019.0475
[6] 孙美丽, 史彩华, 肖本青, 等. 烟草靶斑病叶际微生物群落结构与多样性分析[J]. 烟草科技, 2023, 56(4): 1-9. doi: 10.16135/j.issn1002-0861.2022.0891SUN Meili, SHI Caihua, XIAO Benqing, et al. Composition and diversity of phyllospheric microbial community in tobacco leaves infected by tobacco target spot disease[J]. Tobacco Science & Technology, 2023, 56(4): 1-9. doi: 10.16135/j.issn1002-0861.2022.0891
[7] 刘亭亭, 汪汉成, 孙美丽, 等. 波尔多液对烟草叶际微生物群落结构与代谢功能的影响[J]. 农药学学报, 2022, 24(6): 1446-1455.LIU Tingting, WANG Hancheng, SUN Meili, et al. Effects of Bordeaux mixture on the community structure and metabolic function of tobacco phyllosphere microorganisms[J]. Chinese Journal of Pesticide Science, 2022, 24(6): 1446-1455.
[8] 向立刚, 汪汉成, 罗飞, 等. 感染青枯病与黑胫病烟株的根际土壤、根及茎秆微生物代谢特征分析[J]. 烟草科技, 2023, 56(3): 17-24. doi: 10.16135/j.issn1002-0861.2022.0394XIANG Ligang, WANG Hancheng, LUO Fei, et al. Metabolic characteristics of microorganisms in rhizosphere soil, roots, and stalks of tobacco plants infected with bacterial wilt and black shank[J]. Tobacco Science & Technology, 2023, 56(3): 17-24. doi: 10.16135/j.issn1002-0861.2022.0394
[9] 俞艳玲, 邢旺兴, 陈士景. 7种红曲霉的扫描电镜观察[J]. 浙江中医学院学报, 2005(1): 78-79.YU Yanling, XING Wangxing, CHEN Shijing. Study on the physiological characteristics of seven monascus fungi by scanning electron microscope[J]. Journal of Zhejiang College of Traditional Chinese Medicine, 2005(1): 78-79.
[10]Dai Y F, Wu X M, Wang H C, et al. Spatio-temporal variation in the phyllospheric microbial biodiversity of Alternaria alternata-infected tobacco foliage[J]. Frontiers in Microbiology, 2022, 13: 920109.
[11] 吴盼云, 晁群芳, 赵亚光, 等. 克拉玛依石油污染土壤微生物群落结构及其代谢特征[J]. 基因组学与应用生物学, 2019, 38(5): 2062-2069.WU Panyun, CHAO Qunfang, ZHAO Yaguang, et al. Microbial community structure and metabolic characteristics of oil-contaminated soil in Karamay[J]. Genomics and Applied Biology, 2019, 38(5): 2062-2069.
[12]Zhang L, Wang Y, Xia X Y, et al. Altered gut microbiota in a mouse model of Alzheimers disease[J]. Journal of Alzheimer's Disease, 2017, 60(4): 1241-1257.
[13]Lu L H, Yin S X, Liu X, et al. Fungal networks in yield-invigorating and -debilitating soils induced by prolonged potato monoculture[J]. Soil Biology and Biochemistry, 2013, 65: 186-194.
[14]Magoč T, Salzberg S L. FLASH: Fast length adjustment of short reads to improve genome assemblies[J]. Bioinformatics, 2011, 27(21): 2957-2963.
[15]Edgar R C, Haas B J, Clemente J C, et al. UCHIME improves sensitivity and speed of chimera detection[J]. Bioinformatics, 2011, 27(16): 2194-2200.
[16]Li M J, Shao D T, Zhou J C, et al. Signatures within esophageal microbiota with progression of esophageal squamous cell carcinoma[J]. Chinese Journal of Cancer Research, 2020, 32(6): 755-767.
[17] 陈焘, 周玮, 李宏光, 等. 烟草野火病的发生及综合防治研究进展[J]. 基因组学与应用生物学, 2018, 37(1): 469-476.CHEN Tao, ZHOU Wei, LI Hongguang, et al. Research progress in disease occurrence and integrated control of tobacco wildfire[J]. Genomics and Applied Biology, 2018, 37(1): 469-476.
[18]Lee N M, Lee B H. Thermodynamics on the micellization of pure cationic(DTAB, TTAB, CTAB), nonionic(Tween-20, Tween-40, Tween-80), and their mixed surfactant systems[J]. Journal of the Korean Applied Science and Technology, 2013, 30(4): 679-687.
[19]Suksiri P, Ismail A, Sirirattanachatchawan C, et al. Enhancement of large ring cyclodextrin production using pretreated starch by glycogen debranching enzyme from Corynebacterium glutamicum[J]. International Journal of Biological Macromolecules, 2021, 193(PA): 81-87.
[20]Wu N, Wu X Y, Zhang M Y, et al. Metabolic engineering of Aspergillus niger for accelerated malic acid biosynthesis by improving NADPH availability[J]. Biotechnology Journal, 2024, 19(5): e2400014.
[21]Groza N V, Yarkova T A, Gessler N N, et al. Synthesis of benzoic acid esters and their antimicrobial activity[J]. Pharmaceutical Chemistry Journal, 2024, 58(4): 625-630.
[22] 吴小军, 汪汉成, 孙美丽, 等. 烟草角斑病叶际微生物群落结构与多样性分析[J]. 烟草科技, 2023, 56(10): 1-10. doi: 10.16135/j.issn1002-0861.2023.0282WU Xiaojun, WANG Hancheng, SUN Meili, et al. Composition and diversity of phyllosphere microbial community on tobacco leaves infected with angular leaf spot[J]. Tobacco Science & Technology, 2023, 56(10): 1-10. doi: 10.16135/j.issn1002-0861.2023.0282
[23]Si H Y, Cui B, Liu F, et al. Microbial community and chemical composition of cigar tobacco (Nicotiana tabacum L. ) leaves altered by tobacco wildfire disease[J]. Plant Direct, 2023, 7(12): e551.
[24] 刘宇星, 董醇波, 邵秋雨, 等. 叶际微生物与植物健康研究进展[J]. 微生物学杂志, 2022, 42(2): 88-98.LIU Yuxing, DONG Chunbo, SHAO Qiuyu, et al. Advances on phyllosphere microorganisms and their association with plant health[J]. Journal of Microbiology, 2022, 42(2): 88-98.
[25] 江艳, 桑维钧, 曾尔玲, 等. 烟草茎点病在贵州省的发生及病原鉴定[J]. 江苏农业科学, 2018, 46(10): 92-95.JIANG Yan, SANG Weijun, ZENG Erling, et al. Identification of pathogenic fungus causing tobacco black spot stalk in Guizhou Province[J]. Jiangsu Agricultural Sciences, 2018, 46(10): 92-95.
[26]Wei L L, Chen B, Li J W, et al. Resistance mechanism of Phomopsis longicolla to fludioxonil is associated with modifications in PlOS1, PlOS4 and PlOS5[J]. Pesticide Biochemistry and Physiology, 2024, 201: 105862.
[27]Eduardo L A O D, Antonio P L, Antonio J L, et al. Addressing coffee crop diseases: forecasting Phoma leaf spot with machine learning[J]. Theoretical and Applied Climatology, 2023, 155(3): 2261-2282.
[28]Hansen B C, Gilley M A, Berghuis B G, et al. Effect of fungicide and timing of application on management of Phoma black stem of cultivated sunflowers in the United States[J]. Plant Disease, 2024, 108(7): 2017-2026.
[29] 李纪潮, 张金渝, 蔡明姬, 等. 草果叶斑病类致病菌对新型低毒药剂的敏感性及交互抗性[J]. 南方农业学报, 2022, 53(8): 2153-2160.LI Jichao, ZHANG Jinyu, CAI Mingji, et al. Sensitivity and cross-resistance of pathogen of Amomum tsaoko leaf spot to new low-toxic fungicides[J]. Journal of Southern Agriculture, 2022, 53(8): 2153-2160.
相关知识
烟草根腐病发病与健康根际微生物生物群落分析及球毛壳菌NP2的生防效果研究
炭疽病发病草莓与健康草莓根际细菌群落结构及功能差异
烟草微生物组研究取得新进展
科研丨复旦: 季节性变化驱动水生根际微生物群落结构和功能特性的变化(国人佳作)
《自然》:“叶圈”健康有多重要?中美科学家联合发表植物叶际微生物群领域的新进展
茶树叶面真菌与内生真菌群落结构的比较
微生物多样性与人类健康.docx
热带植物的结构与生态.pptx
健康广藿香与患病广藿香根际土壤微生物群落的结构
药用大黄黑粉病发病部位及根际土壤微生物群落多样性研究
网址: 烟草野火病叶际微生物群落结构多样性与碳源代谢表征 https://www.trfsz.com/newsview1904799.html
推荐资讯
- 1发朋友圈对老公彻底失望的心情 12775
- 2BMI体重指数计算公式是什么 11235
- 3补肾吃什么 补肾最佳食物推荐 11199
- 4性生活姿势有哪些 盘点夫妻性 10428
- 5BMI正常值范围一般是多少? 10137
- 6在线基础代谢率(BMR)计算 9652
- 7一边做饭一边躁狂怎么办 9138
- 8从出汗看健康 出汗透露你的健 9063
- 9早上怎么喝水最健康? 8613
- 10五大原因危害女性健康 如何保 7828